Our Science
Transformative Potential
Approximately 90% of disease-causing proteins are not drugged, or are inadequately drugged, due to limitations with traditional small molecules that target the active site of a protein.
Allostery holds the key to drugging that 90%.
Atavistik Bio is developing precision allosteric therapeutics that have the potential to transform the lives of patients with significant unmet medical need.

———
Leveraging Allostery to Drug the Undruggable
We are harnessing the power of allostery to address unmet patient needs.
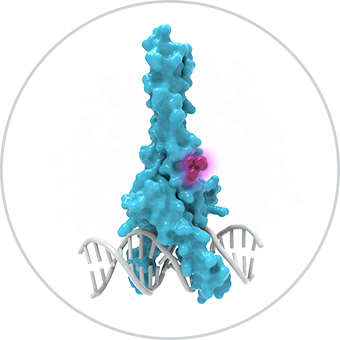
Allostery holds the potential to drug intractable targets
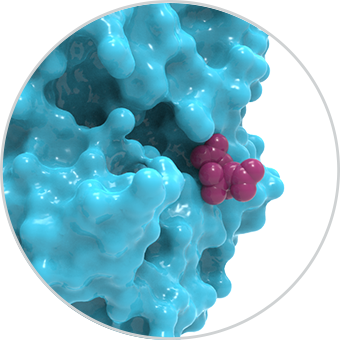
The identification of allosteric pockets fast-tracks discovery of exquisitely selective and highly potent small molecule therapeutics
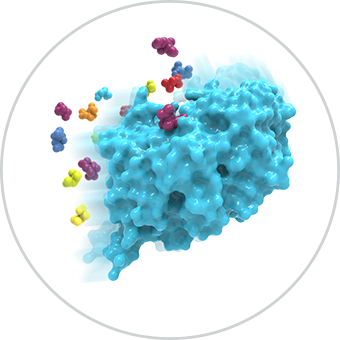
Our platform leverages metabolites to identify functional allosteric pockets across a broad range of target classes
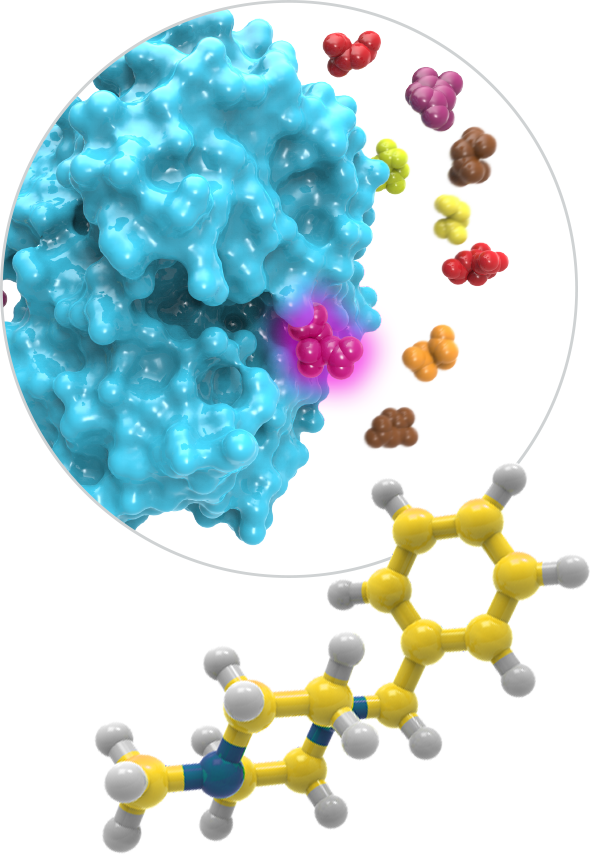
By leveraging the body’s natural regulators, we efficiently and systematically uncover functional allosteric pockets across a broad range of target classes that are not detectable by other techniques, thereby accelerating the development of precision allosteric therapeutics.
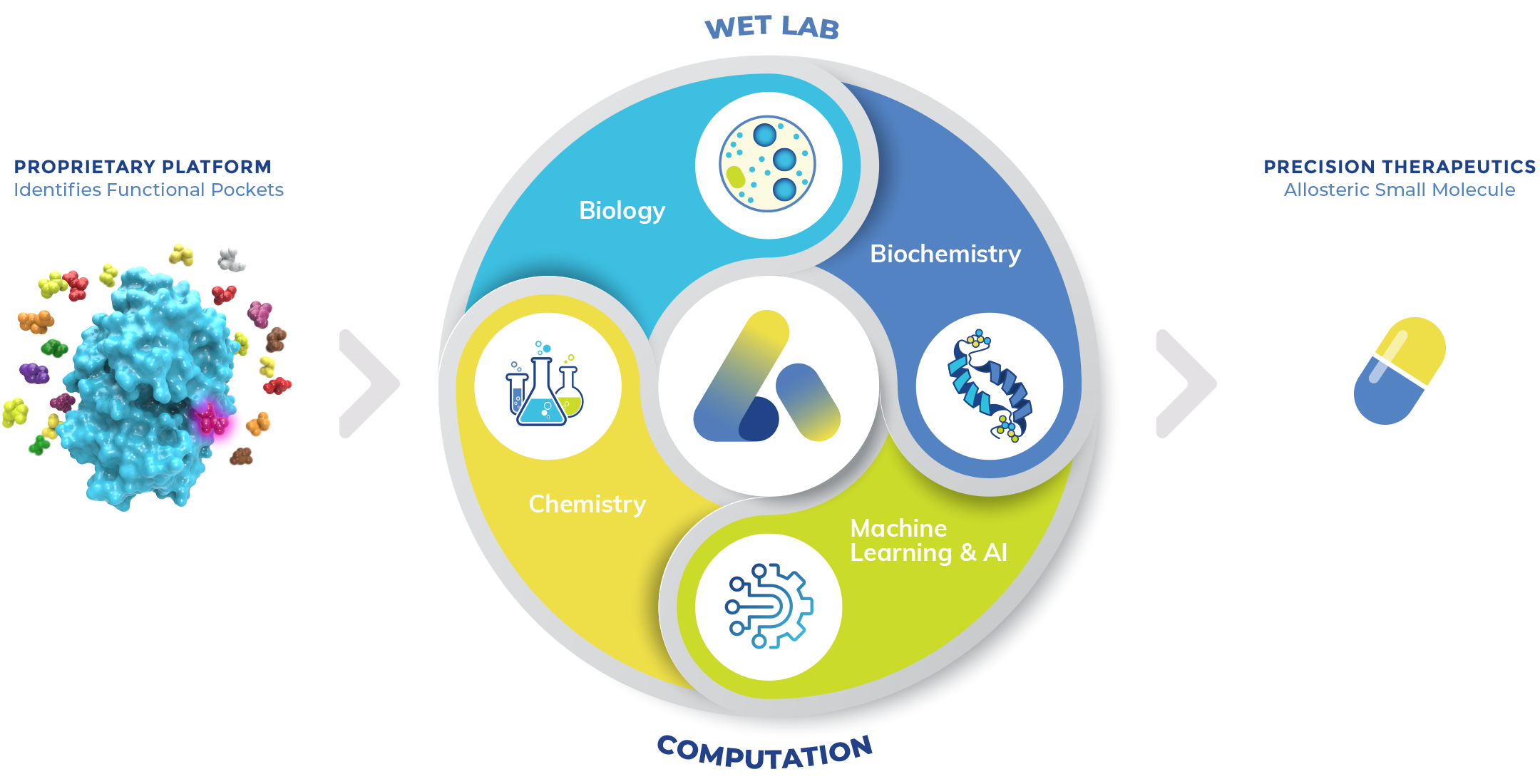
Our Platform
Metabolites modulate the activity of proteins in the human body, often allosterically. Atavistik Bio’s proprietary platform leverages the function of these natural protein regulators as our starting point. By using metabolites as bait, we can pinpoint precisely how and where they bind to proteins to elicit activity, revealing functional pockets for a broad range of target classes. We then apply our AI-enabled structure-based drug discovery engine to design small molecules, enabling the discovery of novel precision allosteric therapeutics. Our highly integrated team and automated data infrastructure makes rapid iteration and progression of chemistry possible.
Therapeutic Approach
We have successfully adapted our platform to screen and identify functional pockets on a broad range of protein classes. While our internal pipeline is focused on high value opportunities in rare diseases and oncology, the versatility of our platform allows us to accelerate the development of groundbreaking medicines across any therapeutic area.
We plan to unlock the broad potential of our platform in other therapeutic areas through strategic partnerships. By combining the disease specific insights of our partners with the capabilities here at Atavistik, we hope to advance transformative therapies in a broad range of therapeutic areas.